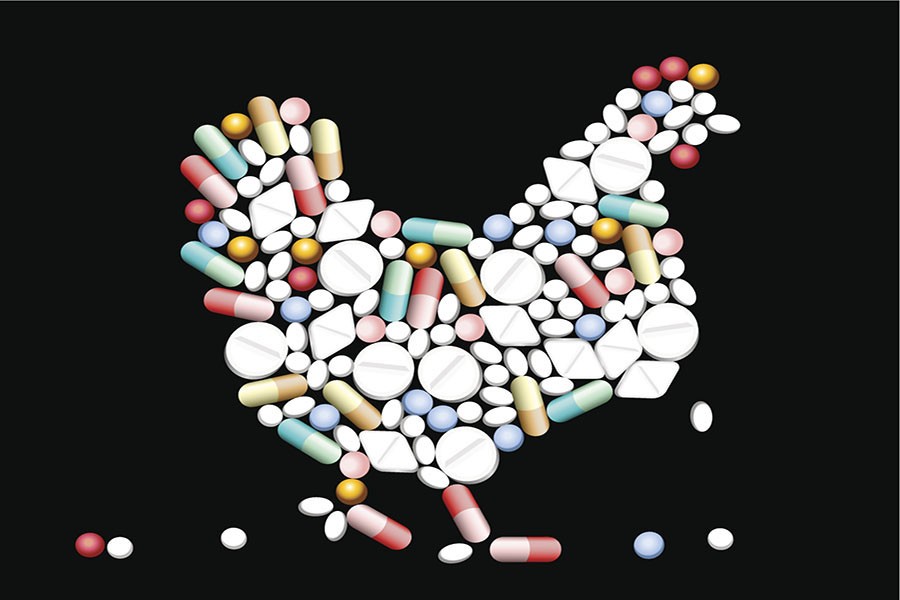
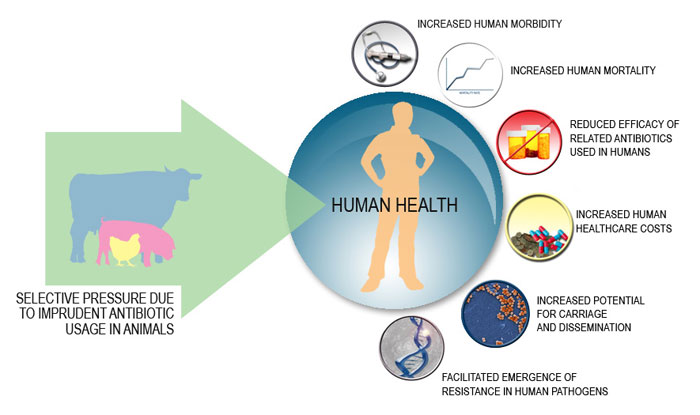
For years, alternative health doctors and health enthusiasts have been touting the detrimental effects from consumption of animals treated with antibiotics. Recently the American Academy of Pediatrics (AAP) took a stand against the use of nontherapeutic antibiotic use in animals citing the potential threat to children’s health, as well as to the public at large,
The AAP technical report, which was published simultaneously in Pediatrics, described how the use of antibiotics in livestock as growth stimulants, and not for treating illnesses, contributes to the threat of antimicrobial resistance and potential infection through the food supply — especially among young children who are most vulnerable to infection.
Jerome Paulson, MD, of the Committee on Environmental Health and co-author of the report, staes that while this is the first report the AAP has issued on the subject, the organization has been engaged in discussions with government agencies and agriculture interests. But he added there has not been much progress on the issue.
“It’s a document that we as pediatricians and the Academy as an organization can use to make the argument to stop the large-scale use of antibiotics in animal feed and water and decrease the use of antibiotics in animals for growth promotions and other nontherapeutic purposes,” he said.
Pediatricians play such an important role because children under 5 have the highest incidence of most food-related infections. Children can become infected through food, contact with animals, and environmental exposures such as when animal runoff contaminates surface waters used for drinking and recreation.
According to the CDC’s Foodborne Diseases Active Surveillance Network, three leading causes of pediatric food-related illness are from exposure to:
- Non-typhoid Salmonella (which can infect fetuses via maternal exposure)
- Campylobacter species
- Staphylococcus aureus
More alarmingly, growing proportions of Salmonella and Campylobacter infections are drug-resistant. In 2013, almost 25% of Campylobacter species were resistant to at least one antibiotic — a dramatic increase from 13% in 1997. Of the 310,000 drug-resistant infections, 23% were resistant to ciprofloxacin and 2% resistant to erythromycin (which, with azithromycin, is considered the preferred antibiotic to treat children with Campylobacter).
Of the 100,000 Salmonella infections, 3% were resistant to ceftriaxone, the first-line pediatric therapy for these organisms. Some Salmonella strains have been found to be resistant to five or more classes of antibiotics.
Paulson says that clinicians should talk to patients and families about purchasing antibiotic-free meat and poultry, which can protect their patients, as well as being aware of their own purchasing choices.
“Clinicians bear some responsibility for this problem because we ourselves are not always prudent in our use of antibiotics,” he said. “Unfortunately, too many people still prescribe antibiotics for colds or sore throats without having an appropriate bacterial diagnosis. So, since clinicians are part of the problem, they can be part of the solution.”
The statement authors wrote that overprescribing of antibiotics is common in the agriculture and food business, and that unlike with humans, these drugs can be given to food animals without a prescription or veterinary oversight. They estimate that approximately 60% of antimicrobial agents sold for use in food animals are also considered important in human medicine. But antibiotics for use in animals is a substantial industry, with more than 32.2 million pounds of antimicrobial drug active ingredients sold for use in animals compared to 7.25 million pounds sold for use in humans.
Paulson said that this issue will ultimately be resolved by either the FDA and the Department of Agriculture or by the marketplace, with large purchasers of meat and poultry telling agricultural business that they will not buy products that have been treated with antibiotics.
“The Academy has been involved in meetings in the past with [these organizations] and we will continue to participate in those meetings,” he concluded. “Now we’ve got a clear document that’s been vetted throughout the Academy and approved by our Board of Directors, so we can perhaps speak with a little more force and share our ideas by sharing the technical report to a wider audience.”
Read More at MedPage Today