In spring 2012, life turned unbelievably miserable for Thomas Stutz. First the former Los Angeles attorney’s wife of 43 years passed away, ending what he calls “a beautiful marriage.” On top of that terrible loss, Stutz was deep in a battle with cancer, and he was losing.
 The father of three adult children and grandfather of six received a diagnosis of stage-2b melanoma in October 2009. Less than two years later, a chest x-ray revealed a lung tumor, pushing his disease to stage-4. Soon after that, the melanoma spread to his liver, spine and shoulder. Thomas tried a range of treatments, but nothing slowed down the disease. This still-active man—who loves biking, hiking, tennis and travel—needed a miracle.
The father of three adult children and grandfather of six received a diagnosis of stage-2b melanoma in October 2009. Less than two years later, a chest x-ray revealed a lung tumor, pushing his disease to stage-4. Soon after that, the melanoma spread to his liver, spine and shoulder. Thomas tried a range of treatments, but nothing slowed down the disease. This still-active man—who loves biking, hiking, tennis and travel—needed a miracle.
Just two weeks after his wife died, Stutz started a treatment called pembrolizumab at the University of California, Los Angeles’ Jonsson Comprehensive Cancer Center. This so-called immunotherapy taught Stutz’s own immune cells to fight his disease. It started out rough. As he recalls, “During the first four to five weeks on the treatment, my symptoms were at their worst level.” He adds, “I had major difficulty breathing and was on oxygen 24/7, confined to a wheelchair and had lost over 25 pounds.” That was more than 15 percent of his usual 155 pounds. But then things started to improve.
“After about five or six weeks on the treatment,” Stutz says, “my breathing started to become a little easier.” Over the next half a dozen months, he felt even better. His appetite returned and his breathing kept improving. “By June of 2012,” he says, “I no longer need the oxygen. By August, I no longer used a wheelchair. By the end of 2012, I was walking up to three miles at a time. By June of 2013, I was back to playing two or three sets of doubles tennis at a time and could ride my bike again.”
Now, three years after his first treatment with pembrolizumab, Stutz says, “I have resumed all of my pre-melanoma activities.” He gets a treatment every three weeks, but with only minimal side effects, and it keeps the tumors in his liver and lung knocked down to just 15 percent of their biggest sizes. Even better, his tumors have remained stable for the past 18 months or so.
Bacteria to Breakthrough
In 1891, William B. Coley—a surgeon at New York Cancer Hospital, which is now the Memorial Sloan Kettering Cancer Center—tested injections of heat-treated bacteria to fight cancer, because some earlier evidence suggested that tumors got smaller in some patients with bacterial infections. In a few cases, Coley’s technique put tumors in complete remission. Presumably, the bacteria turned on the person’s immune system, which fought the bacteria and the cancer.
Unfortunately, Coley’s results proved too inconsistent, and this approach was left un-pursued for decades. Nonetheless, cancer researchers have considered other approaches to immunotherapy in the intervening years; some scientists thought that it offered great potential, and others considered it a complete dead end. As Christopher Parish of the John Curtin School of Medical Research at the Australian National University in Canberra wrote in a 2003 issue of Immunology and Cell Biology, “One of the most controversial issues in immunology for over a century has been whether an effective immune response can be elicited against malignant tumours.” He continued, “Whether the immunology community has believed cancer immunotherapy is feasible or impossible has been largely determined by the prevailing immunological paradigms at that time. In fact, during the last 110 years it is possible to trace at least five dramatic fluctuations in attitude towards cancer immunotherapy.”
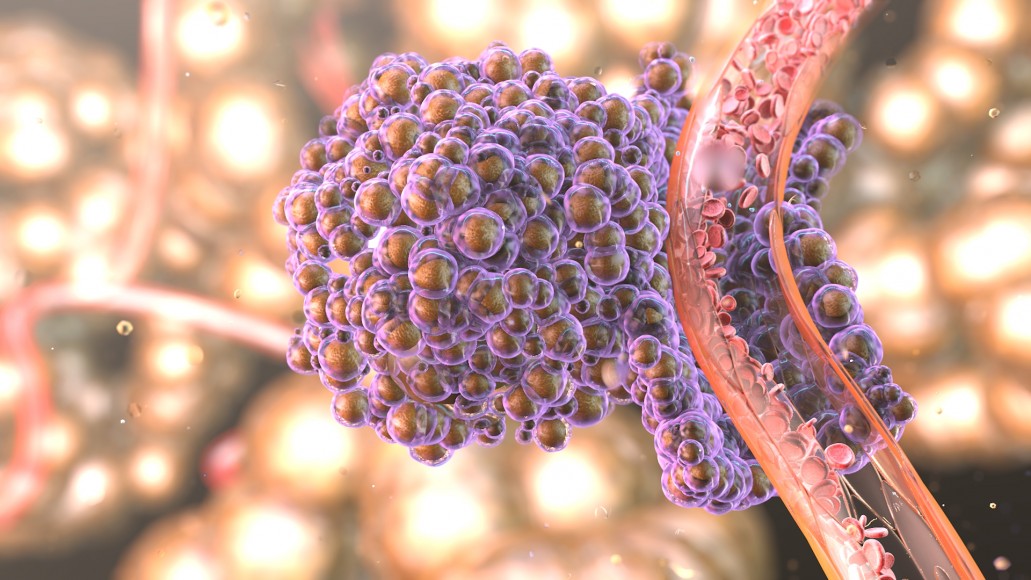
Now, though, advances in immunotherapy are changing cancer treatment and turning some deadly diseases into manageable ones. In fact, in 2013, Science magazine selected immunotherapy as the “Breakthrough of the Year”. The article describing this advance points out contributions from scientists in academics—some of them described below—and the biopharmaceutical industry.
Controlling the Switches
The human body possesses immune cells called cytotoxic T lymphocytes (CTLs), which can kill cancer cells. To keep the immune system from going overboard and attacking things it shouldn’t, though, a molecule called CTLA-4 can be activated to turn off the immune cell. In 1995, James Allison—then at the University of California, Berkeley, and now at the University of Texas MD Anderson Cancer Center—suggested that inhibiting CTLA-4 could unleash the immune system to fight cancer.
Allison tested this idea in the lab, and this eventually generated ipilimumab, which was approved by the US Food and Drug Administration (FDA) as a treatment for melanoma in 2011. In patients with this skin cancer, ipilimumab turns off the mechanism that normally slows down the immune system.
When asked how this treatment impacts melanoma, Paolo Antonio Ascierto of the Unit of Medical Oncology and Innovative Therapies at the Istituto Nazionale per lo Studio e la Cura dei Tumori in Napoli, Italy, says, “My opinion is that—as for melanoma and for all the other cancers—the goal is to make chronic the disease. With the immunotherapy this is now really possible.”
The thinking behind CTLA-4-based approaches is leading to other techniques. For instance, another immune-system “brake” provides a second way to slow down cancer. A molecule called programmed cell death protein 1 (PD-1) appears on the surface of many of the immune system’s T cells. This protein can turn on a system that causes the T cells to self-destruct. So much like the CTLA-4 mechanism, PD-1 can slow down the immune system’s fight against cancer. In fact, some tumor cells even cover themselves in molecules that turn on PD-1 in T cells, causing the T cells to kill themselves instead of the tumor.
Learning more about this system, in conjunction with the success of ipilimumab, spawned a wide range of drug research on forms of immunotherapy that would block PD-1. In fact, many pharmaceutical companies have PD-1-blocking drugs in development or on the market. For example, the drug that keeps fending off Thomas Stutz’s melanoma—pembrolizumab—is an anti-PD-1 therapy.
At the American Society of Hematology’s 2014 annual meeting in San Francisco, Merck reported promising results of using its pembrolizumab against classical Hodgkin lymphoma and advanced triple-negative breast cancer, which is one of the most dangerous forms of this disease.
Subduing the Side Effects
The great benefits of immunotherapies based on blocking CTLA-4 and PD-1, though, do not come without side effects. These treatments can generate inflammatory reactions caused by the immune system getting cranked up a bit too much.
This inflammation can appear as skin rashes and intestinal trauma. For the most part, adding an anti-inflammatory drug to the treatment takes care of the problem. Also, the inflammation issues tend to be worse with CTLA-4 treatments than PD-1 inhibitors. Although it might seem like adding an anti-inflammatory would reduce the efficacy of the checkpoint inhibitors, this treatment could actually improve the outcome, because data show that nonsteroidal anti-inflammatory drugs (NSAIDs) enhance the effects of fighting tumors.
The side effects can be even more troublesome in combinations of immunotherapy. For example, Jedd Wolchok of the Memorial Sloan Kettering Cancer Center and Mario Sznol of Yale University combined ipilimumab and nivolumab, a PD-1 blocker, as a treatment for people with metastatic melanoma. The patients suffered more side effects than either drug alone would cause, but an added anti-inflammatory controlled the trouble. Plus, this combination treatment turned out to be worth any side-effect snag, because it cut tumor size in half for 50 percent of the patients. Although that result came from a small study that needs to be replicated with more patients, this outcome would make almost anyone with advanced melanoma ready to take on the side effects, especially since they can be so easily controlled.
CAR T’ing Away Cancer
Some of the most exciting applications of immunotherapy for cancer come from engineered T cells, called chimeric antigen receptor T cells, or CAR T cells. Here, scientists modify a person’s own T cells to fight a specific cancer.
In the 27 June 2013 issue of Nature, Bruce Levine and Carl H. June—both at the University of Pennsylvania’s Perelman School of Medicine—described using CAR T cells to “eradicate kilograms of leukaemia in a few weeks.” Likewise, in the 16 October 2014 New England Journal of Medicine, June and his colleagues described a CAR-based technique that battled acute lymphoblastic leukemia into remission in 90 percent of the patients.
In general, the applications of this approach to immunotherapy keep expanding. At the 2014 American Society of Hematology meeting, a collection of biotechnology and pharmaceutical companies plus academic researchers reported other advances with CAR T cells against leukemias and lymphomas.
Most of today’s immunotherapies for cancer are in the experimental stage—in clinical trials at best. As a growing number of studies show the benefits of these approaches, more of these treatments should become available—and to patients at earlier stages of disease. That way, people can get these advanced treatments before suffering through less effective options or letting the disease accelerate and spread. In addition, some combinations—such as immunotherapy plus radiation or traditional chemotherapy—might prove even more effective at fighting cancer.
Existing techniques are already changing lives. Without immunotherapy, Stutz says, “I probably would not have survived the spring of 2012. With it, I have my normal life back.”
Source: Sciam Benchmarks